What is KHINDIVI?

Depiction only. Not actual label.
KHINDIVI™ (hydrocortisone) 1 mg/mL oral solution is the only FDA-approved oral hydrocortisone solution for pediatric patients aged 5 years and older with adrenocortical insufficiency who require daily hormone replacement therapy.1
Limitation of Use: KHINDIVI is not approved for increased dosing during periods of stress or acute events. Use a different hydrocortisone-containing drug product for stress dosing.1
Hydrocortisone dosing challenges
Hydrocortisone dosing inaccuracies can occur through pill manipulation or compounded formulations.
Crushing or splitting tablets may lead to imprecise measurements and inconsistent product dimensions. Compounded formulations pose additional risks because they lack FDA approval, and quality standards vary depending on the compounding facility. These factors may result in under- or overdosing.2-4
Dosing challenges at home3
In a study that examined dose variation in hydrocortisone tablets split into quarters by different operators
>50%
of doses could not be achieved* by quartering tablets
*In a study of quartered 10-mg hydrocortisone tablets, 54% (43 out of 80 tablets) failed to achieve ±10% of the target 2.5-mg dose.
Compounding at the pharmacy4
Issues with hydrocortisone suspensions produced by pharmacy compounding
21%
of batches lacked uniformity of net mass or drug content*
4%
of batches completely failed to contain any trace of the labeled drug
*Evaluation of 56 batches containing 1124 capsules. Capsule samples collected randomly from volunteering caregivers.
Other dosing challenges
- Patients who can’t swallow tablets or need special administration (like a gastric tube) may face inconsistent dosing from compounding
- Children needing very small doses (under 0.5 mg) may receive inaccurate amounts when tablets are cut or crushed2
- Compounded pharmacy suspensions aren’t FDA-approved and may have inconsistent composition, creating dosing uncertainty4
These limitations highlight the need for accurate and reliable options in pediatric adrenal care.
KHINDIVI helps ensure dosing accuracy
KHINDIVI offers the advantages of oral liquid administration5

Eliminates the need for compounding and splitting or crushing tablets

Designed to help provide simple dosing as prescribed

Does not require mixing or shaking

Stable up to 120 days at room temperature
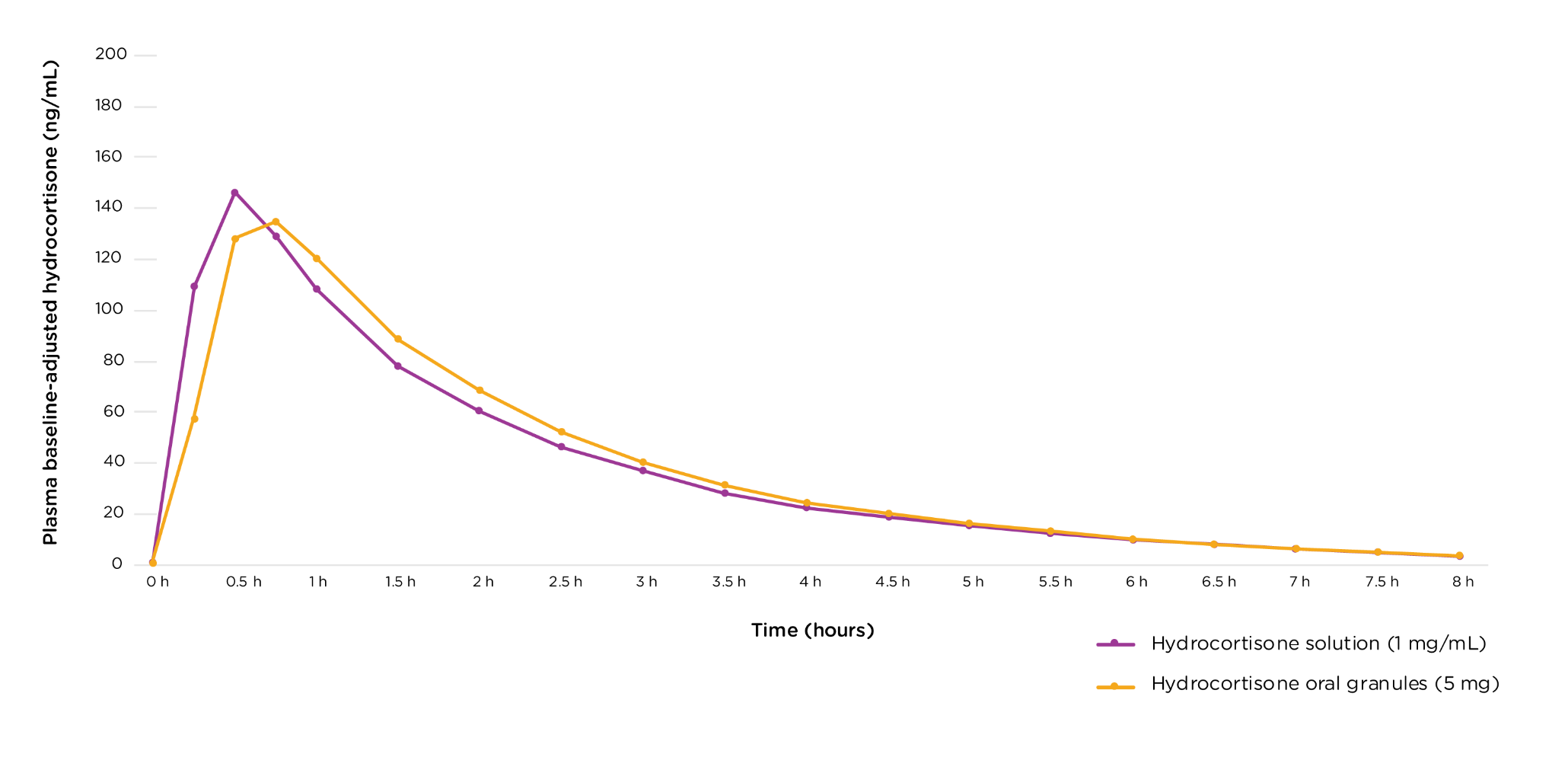
Safety and efficacy based on bioequivalence to ALKINDI SPRINKLE® (hydrocortisone) oral granules6
The study showed that under fasted conditions, the pharmacokinetics of KHINDIVI were bioequivalent to ALKINDI SPRINKLE and had the same rate and extent of absorption.
See more data on the bioequivalence studies.
No new side effects were observed in this bioequivalence study.6
Click below to submit your prescription now
to Anovo® Specialty Pharmacy
INDICATION
KHINDIVI is a corticosteroid indicated as replacement therapy in pediatric patients 5 years of age and older with adrenocortical insufficiency.
Limitation of Use: KHINDIVI is not approved for increased dosing during periods of stress or acute events. Use a different hydrocortisone-containing drug product for stress dosing.
IMPORTANT SAFETY INFORMATION
Contraindication
Hypersensitivity to hydrocortisone or any of the other ingredients in KHINDIVI oral solution.
Warnings and Precautions
Adrenal Crisis: Undertreatment or sudden discontinuation of therapy with KHINDIVI may lead to symptoms of adrenal insufficiency, adrenal crisis, and death. Adrenal crisis may also be induced by stress events, such as infections or surgery when patients require higher doses of corticosteroids. During periods of stress (e.g., infections, surgery), switch to another oral hydrocortisone product and increase the dose if oral medications are tolerated. Monitor patients when switching to KHINDIVI to ensure KHINDIVI is providing the same level of hydrocortisone exposure as the previously used oral hydrocortisone formulation. If symptoms of adrenal insufficiency occur, increase the total daily dosage of KHINDIVI.
Systemic Adverse Reactions Due to Inactive Ingredients
Hyperosmolarity
KHINDIVI is not approved in pediatric patients less than 5 years of age. The inactive ingredients polyethylene glycol 400, propylene glycol, and glycerin undergo substantial systemic absorption, individually or in combination, resulting in increased plasma osmolarity in all pediatric patients, especially in pediatric patients less than 5 years of age. Monitor pediatric patients using KHINDIVI for signs and symptoms consistent with hyperosmolarity.
Metabolic Acidosis and Other Adverse Reactions
The inactive ingredient polyethylene glycol 400 and propylene glycol that may result in metabolic acidosis, hypoglycemia, hepato-renal injury, and central nervous system toxicity (e.g., seizure and coma), may increase the risk of adrenal crisis. Monitor laboratory values and for physical signs and symptoms of these adverse reactions.
Laxative Effects Due to Inactive Ingredients
The inactive ingredients polyethylene glycol 400 and glycerin, whether alone or in combination, may cause gastrointestinal irritation resulting in vomiting and/or diarrhea. These gastrointestinal reactions may increase the risk of adrenal crisis. Monitor for signs or symptoms of gastrointestinal irritation and associated fluid and electrolyte abnormalities.
Immunosuppression and Increased Risk of Infection With Use of a Dosage Greater Than Replacement: The use of a greater than replacement dosage can suppress the immune system and increase the risk of infection with any pathogen, including viral, bacterial, fungal, protozoan, or helminthic pathogens. Monitor for the development of infection and consider dosage reduction as needed.
Growth Retardation: Long-term use in excessive doses may cause growth retardation. Use the minimum dosage of KHINDIVI to achieve desired clinical response and monitor the patient’s growth.
Cushing’s Syndrome Due to Use of Excessive Doses of Corticosteroids: Prolonged use with supraphysiologic doses may cause Cushing’s syndrome. Monitor patients for signs and symptoms of Cushing’s syndrome every 6 months.
Decrease in Bone Mineral Density: Corticosteroids decrease bone formation and increase bone resorption which may lead to the development of osteoporosis. Use the minimum dosage of KHINDIVI to achieve desired clinical response.
Psychiatric Adverse Reactions: Use may be associated with severe psychiatric adverse reactions, such as euphoria, mania, psychosis with hallucinations and delirium, or depression. Symptoms typically emerge within a few days or weeks of starting the treatment. Most reactions resolve after either dose reduction or withdrawal, although specific treatment may be necessary. Monitor patients for behavioral and mood disturbances during treatment. Instruct caregivers and/or patients to seek medical advice if psychiatric symptoms develop.
Ophthalmic Adverse Reactions: Cataracts, glaucoma, and central serous chorioretinopathy have been reported with prolonged use of high doses. Monitor patients for blurred vision or other visual disturbances, and if they occur, refer them to an ophthalmologist.
Gastrointestinal Adverse Reactions: There is an increased risk of gastrointestinal perforation in patients with certain gastrointestinal disorders. Signs of gastrointestinal perforation, such as peritoneal irritation, may be masked in patients receiving corticosteroids. Corticosteroids should be used with caution if there is a probability of impending perforation, abscess, or other pyogenic infections; diverticulitis, fresh intestinal anastomoses, and active or latent peptic ulcer.
Concurrent administration of corticosteroids with nonsteroidal anti-inflammatory drugs (NSAIDs) may increase the risk of gastrointestinal adverse reactions. Monitor patients receiving corticosteroids and concomitant NSAIDs for gastrointestinal adverse reactions.
Risk of Kaposi’s Sarcoma With Use of a Dosage Greater Than Replacement: Kaposi’s sarcoma has been reported to occur in patients receiving corticosteroid therapy, most often for chronic conditions at a dosage greater than replacement (supraphysiologic dosage). If patients take a supraphysiologic chronic dosage of KHINDIVI, they are at increased risk of developing Kaposi’s sarcoma.
Vaccination: Administration of live vaccines may be acceptable in KHINDIVI-treated pediatric patients with adrenocortical insufficiency who receive replacement corticosteroids.
Adverse Reactions
The serious adverse reactions associated with KHINDIVI are adrenal crisis, systemic adverse reactions due to inactive ingredients, immunosuppression, and increased risk of infection with dosage greater than replacement, Cushing’s Syndrome, growth retardation, Kaposi’s Sarcoma risk, psychiatric, ophthalmic medical and gastrointestinal adverse reactions.
To report a suspected adverse event related to KHINDIVI, contact Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at http://www.fda.gov/MedWatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.
INDICATION
KHINDIVI is a corticosteroid indicated as replacement therapy in pediatric patients 5 years of age and older with adrenocortical insufficiency.
Limitation of Use: KHINDIVI is not approved for increased dosing during periods of stress or acute events. Use a different hydrocortisone-containing drug product for stress dosing.
IMPORTANT SAFETY INFORMATION
Contraindication
Hypersensitivity to hydrocortisone or any of the other ingredients in KHINDIVI oral solution.
Warnings and Precautions
Adrenal Crisis: Undertreatment or sudden discontinuation of therapy with KHINDIVI may lead to symptoms of adrenal insufficiency, adrenal crisis, and death. Adrenal crisis may also be induced by stress events, such as infections or surgery when patients require higher doses of corticosteroids. During periods of stress (e.g., infections, surgery), switch to another oral hydrocortisone product and increase the dose if oral medications are tolerated. Monitor patients when switching to KHINDIVI to ensure KHINDIVI is providing the same level of hydrocortisone exposure as the previously used oral hydrocortisone formulation. If symptoms of adrenal insufficiency occur, increase the total daily dosage of KHINDIVI.
Systemic Adverse Reactions Due to Inactive Ingredients
Hyperosmolarity
KHINDIVI is not approved in pediatric patients less than 5 years of age. The inactive ingredients polyethylene glycol 400, propylene glycol, and glycerin undergo substantial systemic absorption, individually or in combination, resulting in increased plasma osmolarity in all pediatric patients, especially in pediatric patients less than 5 years of age. Monitor pediatric patients using KHINDIVI for signs and symptoms consistent with hyperosmolarity.
Metabolic Acidosis and Other Adverse Reactions
The inactive ingredient polyethylene glycol 400 and propylene glycol that may result in metabolic acidosis, hypoglycemia, hepato-renal injury, and central nervous system toxicity (e.g., seizure and coma), may increase the risk of adrenal crisis. Monitor laboratory values and for physical signs and symptoms of these adverse reactions.
Laxative Effects Due to Inactive Ingredients
The inactive ingredients polyethylene glycol 400 and glycerin, whether alone or in combination, may cause gastrointestinal irritation resulting in vomiting and/or diarrhea. These gastrointestinal reactions may increase the risk of adrenal crisis. Monitor for signs or symptoms of gastrointestinal irritation and associated fluid and electrolyte abnormalities.
Immunosuppression and Increased Risk of Infection With Use of a Dosage Greater Than Replacement: The use of a greater than replacement dosage can suppress the immune system and increase the risk of infection with any pathogen, including viral, bacterial, fungal, protozoan, or helminthic pathogens. Monitor for the development of infection and consider dosage reduction as needed.
Growth Retardation: Long-term use in excessive doses may cause growth retardation. Use the minimum dosage of KHINDIVI to achieve desired clinical response and monitor the patient’s growth.
Cushing’s Syndrome Due to Use of Excessive Doses of Corticosteroids: Prolonged use with supraphysiologic doses may cause Cushing’s syndrome. Monitor patients for signs and symptoms of Cushing’s syndrome every 6 months.
Decrease in Bone Mineral Density: Corticosteroids decrease bone formation and increase bone resorption which may lead to the development of osteoporosis. Use the minimum dosage of KHINDIVI to achieve desired clinical response.
Psychiatric Adverse Reactions: Use may be associated with severe psychiatric adverse reactions, such as euphoria, mania, psychosis with hallucinations and delirium, or depression. Symptoms typically emerge within a few days or weeks of starting the treatment. Most reactions resolve after either dose reduction or withdrawal, although specific treatment may be necessary. Monitor patients for behavioral and mood disturbances during treatment. Instruct caregivers and/or patients to seek medical advice if psychiatric symptoms develop.
Ophthalmic Adverse Reactions: Cataracts, glaucoma, and central serous chorioretinopathy have been reported with prolonged use of high doses. Monitor patients for blurred vision or other visual disturbances, and if they occur, refer them to an ophthalmologist.
Gastrointestinal Adverse Reactions: There is an increased risk of gastrointestinal perforation in patients with certain gastrointestinal disorders. Signs of gastrointestinal perforation, such as peritoneal irritation, may be masked in patients receiving corticosteroids. Corticosteroids should be used with caution if there is a probability of impending perforation, abscess, or other pyogenic infections; diverticulitis, fresh intestinal anastomoses, and active or latent peptic ulcer.
Concurrent administration of corticosteroids with nonsteroidal anti-inflammatory drugs (NSAIDs) may increase the risk of gastrointestinal adverse reactions. Monitor patients receiving corticosteroids and concomitant NSAIDs for gastrointestinal adverse reactions.
Risk of Kaposi’s Sarcoma With Use of a Dosage Greater Than Replacement: Kaposi’s sarcoma has been reported to occur in patients receiving corticosteroid therapy, most often for chronic conditions at a dosage greater than replacement (supraphysiologic dosage). If patients take a supraphysiologic chronic dosage of KHINDIVI, they are at increased risk of developing Kaposi’s sarcoma.
Vaccination: Administration of live vaccines may be acceptable in KHINDIVI-treated pediatric patients with adrenocortical insufficiency who receive replacement corticosteroids.
Adverse Reactions
The serious adverse reactions associated with KHINDIVI are adrenal crisis, systemic adverse reactions due to inactive ingredients, immunosuppression, and increased risk of infection with dosage greater than replacement, Cushing’s Syndrome, growth retardation, Kaposi’s Sarcoma risk, psychiatric, ophthalmic medical and gastrointestinal adverse reactions.
To report a suspected adverse event related to KHINDIVI, contact Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at http://www.fda.gov/MedWatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.
References: 1. KHINDIVI Package Insert. Eton Pharmaceuticals, Inc; 2025. 2. US Food and Drug Administration. Pharmacy Compounding of Human Drug Products Under Section 503A of the Federal Food, Drug, and Cosmetic Act: Guidance for Industry. US Department of Health and Human Services. Published June 2016. Accessed July 16, 2025. https://www.fda.gov/media/94393/download 3. Madathilethu J, Roberts M, Peak M, Blair J, Prescott R, Ford JL. Content uniformity of quartered hydrocortisone tablets in comparison with mini-tablets for paediatric dosing. BMJ Paediatr Open. 2018;2(1):e000198. doi:10.1136/bmjpo-2017-000198 4. Neumann U, Burau D, Spielmann S, et al. Quality of compounded hydrocortisone capsules used in the treatment of children. Eur J Endocrinol. 2017;177(2):239-242. doi:10.1530/EJE-17-0248 5. Lajoinie A, Janiaud P, Henin E, et al. Assessing the effects of solid versus liquid dosage forms of oral medications on adherence and acceptability in children. Cochrane Database of Systematic Reviews 2017, Issue 9. Art. No.: CD012783 doi:10.1002/14651858.CD012783 6. Seo BW, Freedman ML, Hoos I, Radosavljevic D, Christensen A. SAT-427 bioequivalence study between 5 mg of hydrocortisone solution (ET-400) versus hydrocortisone oral granules (Alkindi Sprinkle). J Endocr Soc. 2025:9(suppl 1): bvaf149.295. doi.org/10.1210/jendso/bvaf149.295
